Why Combining Agile and Waterfall Improves the Drug Development Process
Traditionally, Healthcare, Pharmaceutical and Life Science companies develop software using a “Waterfall” model. Under the Waterfall model, every stage of development – including product definition, user requirements, designing, coding and testing – takes place in sequence. Testing only happens after coding is complete; coding only happens after the design stage is complete, and so on.
There are many advantages to this model. It is clear, well-documented, organised, and it can make sense for projects whose scope and requirements are predictable from the beginning, with little chance of unexpected developments.
Drug development teams within Pharmaceutical companies tend to be highly specialised and accustomed to working in a rigid and sequential environment. This is especially important due to the regulatory environment healthcare organisations operate under, and to the potential clinical risk associated with patient care.
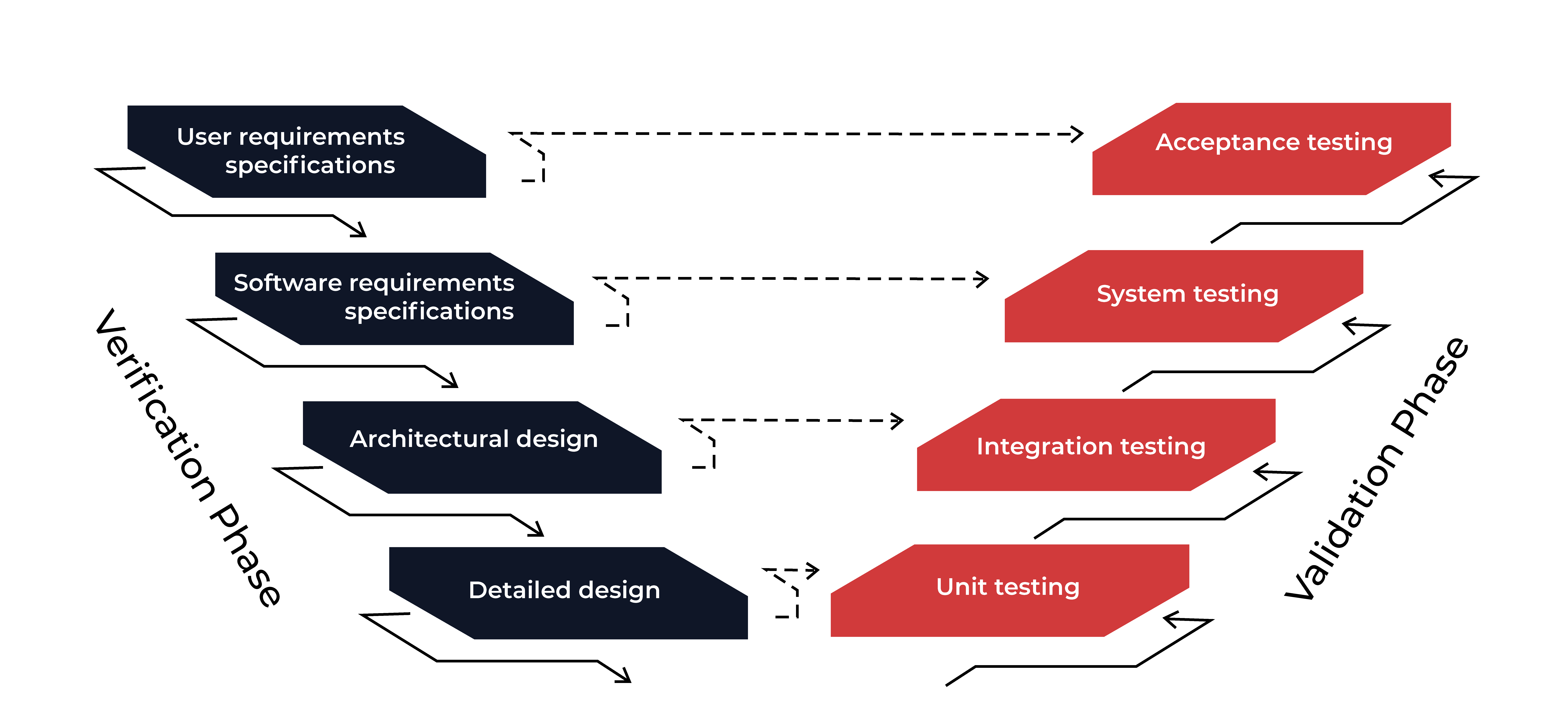
Such structured processes and approaches have typically necessitated a Waterfall model when developing new applications. A stepwise process in which requirements are dictated at the start of a project – including design-control, implementation, and verification – ensures risk is managed throughout the development.
This model is shown in the image below:
Understanding Agile
But in this era of ever-changing external factors, such as regulatory guidelines and new emerging technologies, Healthcare organisations need to be responsive, nimble, and able to seize the opportunity more than ever to be able to pivot their strategies in order to provide the highest level of patient care and patient engagement.
As a result, software developers of Healthcare and medical device applications have increasingly been turning to Agile development practices that have been in use in other industries for many years.
In the world of Healthcare application development, the stepwise waterfall approach can be too simplistic and rigid. Outlining requirements up front means that system updates or changes may not be realised until development is complete. This can create wasted work and delayed timelines. Instead, software developers have embraced the Agile development model, which allows for creative and iterative product development cycles.
In the Agile development process, work is divided into short “sprints”. Instead of designing, coding, and testing the final product in a single round, Agile projects develop the product incrementally. In each sprint, or round of development, a minimal number of new features are designed, coded, and tested. Each sprint has a defined duration (usually in weeks) with a running list of deliverables, planned at the start of the sprint. At the end of each sprint, the team can review and evaluate the results, using the insights gained as feedback for planning the next sprint. This approach is shown in the image below:
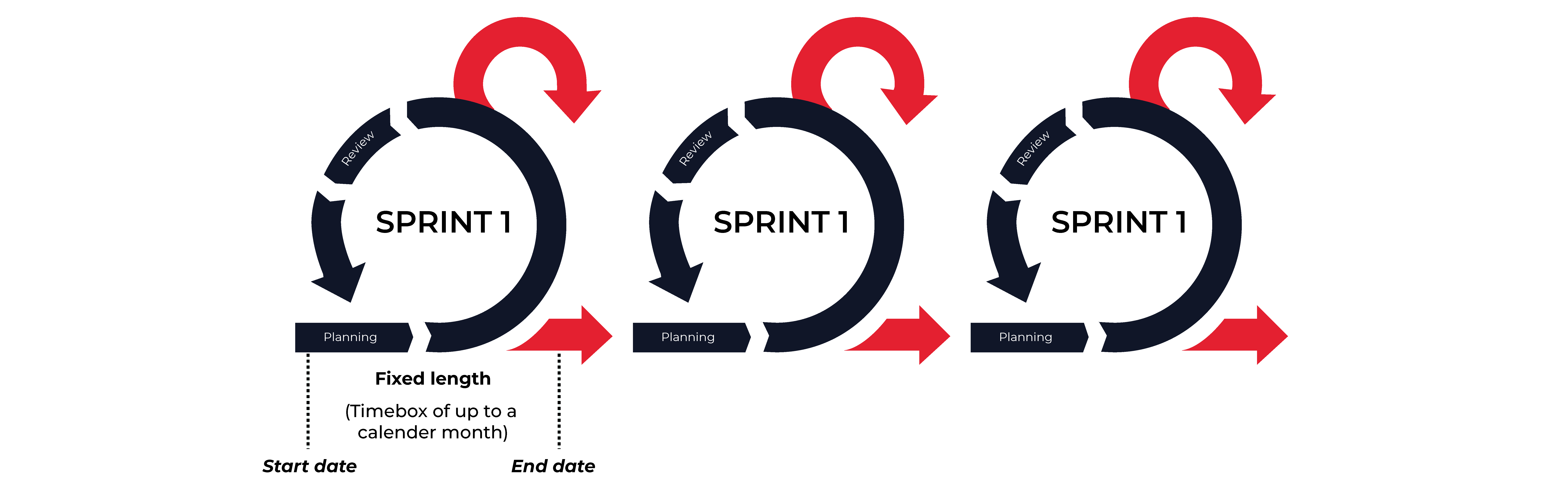
Benefits of Agile
There are many benefits to this short feedback loop:
- Companies can quickly develop Proofs of Concepts and Minimal Viable Products (MVP), which can be crucial for Pharmaceutical companies keen to get products out to market quickly and to collect user feedback during the development cycles to ensure maximum applicability.
- Agile development also means that testing is integrated into every stage of the development process. Reliability and security are never an afterthought. Agile development is especially valuable when working with external development teams. When companies work with external developers, it’s crucial that they feel confident that they are fully aware of, involved in, and in control of the development process.
- Additionally, Agile development allows for maximum transparency. The short development loop means that companies can quickly observe the results of new changes and provide feedback on the product at every stage.
- The increased predictability of Agile development processes also means that outsourcing companies can be more confident about avoiding cost overruns and delays.
Blending Waterfall and Agile Practices for Pharma
At DMI, we specialise in Agile development because it allows us to develop software quickly, transparently, cost-effectively, and securely, while understanding the regulatory environment our clients must work within.
We blend our Agile development practices into Waterfall projects to ensure compliance, traceability, and quality, while embedding flexible agile processes and design thinking to reduce risk, control costs and ensure flexibility through the development cycles.
We have embedded the benefits of both Waterfall and Agile methods into our Healthcare projects and believe this model of delivery ensures regulatory compliance, design control, and adherence to quality management systems. It also allows for adaptability, flexibility and cost control. This blended approach is show in the image below.
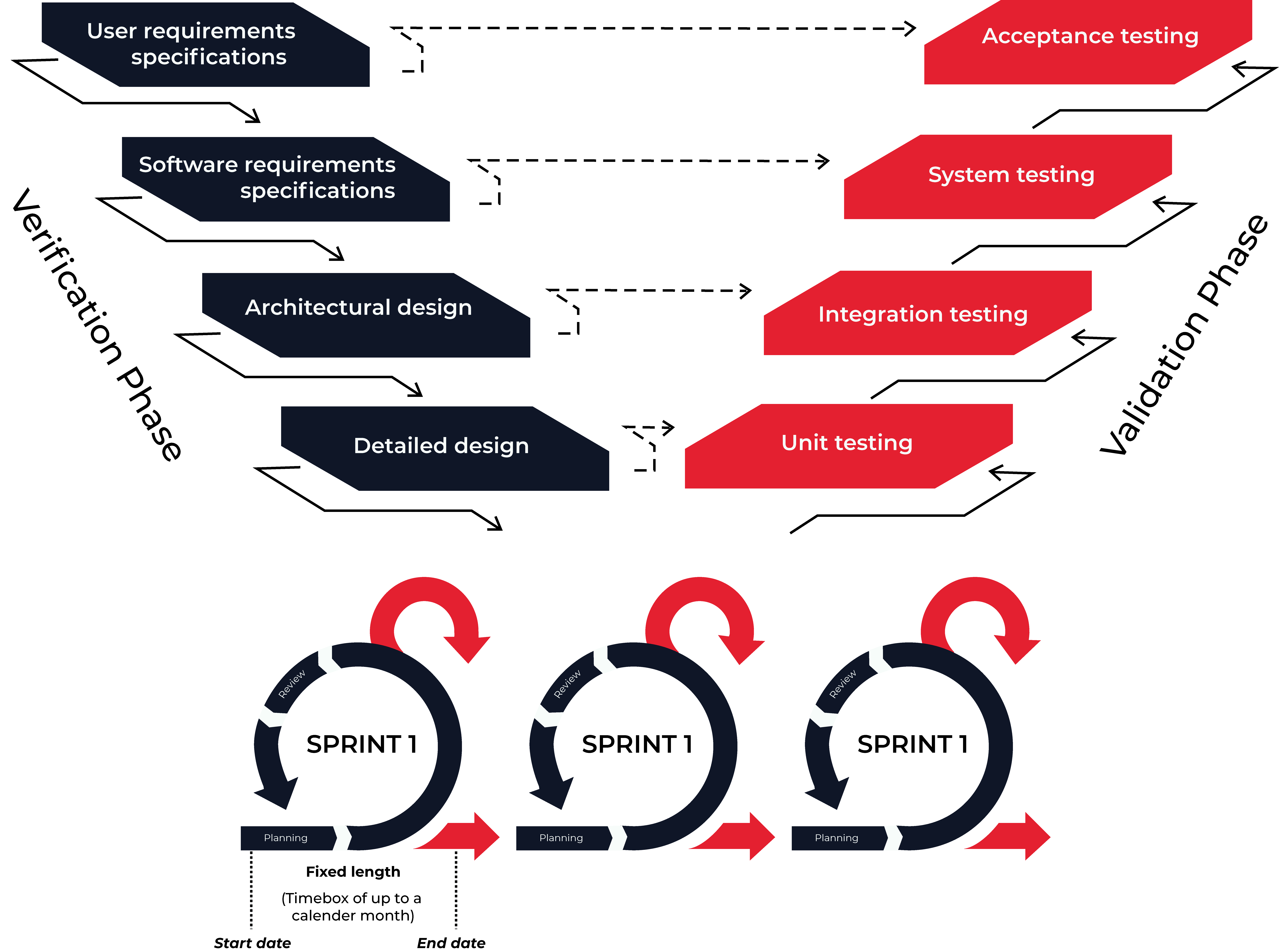
Your Leadership’s Support for a Blended Approach
DMI recognises that company leadership plays an important role in guiding employees towards an embrace of a more Agile mindset. Leaders should define the qualitative and quantitative impact of Agile in terms of time to market drugs, increasing the productivity of R&D teams, and highlighting its role in nurturing innovation.
We can help companies re-imagine the target operating model. Creating a blueprint that identifies areas for Waterfall and Agile methodologies can help create an outline that communicates how this can be translated to the target operating model. This involves reviewing the existing portfolio and team structures to select teams that will benefit from agile and then re-designing the processes.
About DMI’s Healthcare & Life Sciences Practice
DMI’s Healthcare & Life Sciences practice has experience providing digital services to modernise the patient experience, deliver health analytics, and establish health information exchange.
We serve numerous Healthcare clients in the public and private sectors, including the U.S. Department of Health and Human Services, Takeda Pharmaceutical Company, McKesson, Anthem, UnitedHealth, Allergan, St. Luke’s Hospital, Fresenius and Zimmer-Biomet.
We are Healthcare industry experts who are free to think differently and develop Healthcare solutions that empower and inspire.
Connect with us for a discussion about blending Agile and Waterfall methods within your company.


